ACT vs Avalanche -- Intermediate Stage Dry AMD
Sept 13, 2014 19:21:01 GMT
JHam, michaelbrom, and 6 more like this
Post by tradeup on Sept 13, 2014 19:21:01 GMT
After doing some research, it appears Avalanche Biotech (pipeline) is going after the same Intermediate Stage Dry AMD patient population as ACT -- with a candidate due for clinical trials within a few years. ACT's lead investigator on its Ocular Advisory Board, Dr. Steven Schwartz, is a co-founder of Avalanche.
Here's a summary of findings ...
Avalanche's lead candidate, AVA-101, is in a Phase 2a trial targeting Wet-AMD. Results look very promising (AAVL stock has soared since IPO). Top-line results are expected mid-2015 (here's a glimpse at data presented at Euretina 2014). However, AVA-201, in pre-clinical development, is targeting Wet AMD "Prevention." And if AVA-101 is a home run, which it looks like it may be, AVA-201 could see similar success.
Per Avalanche's website:
How it works from Avalanche's S-1 filing:
Why Avalanche's technology could be blockbuster ...
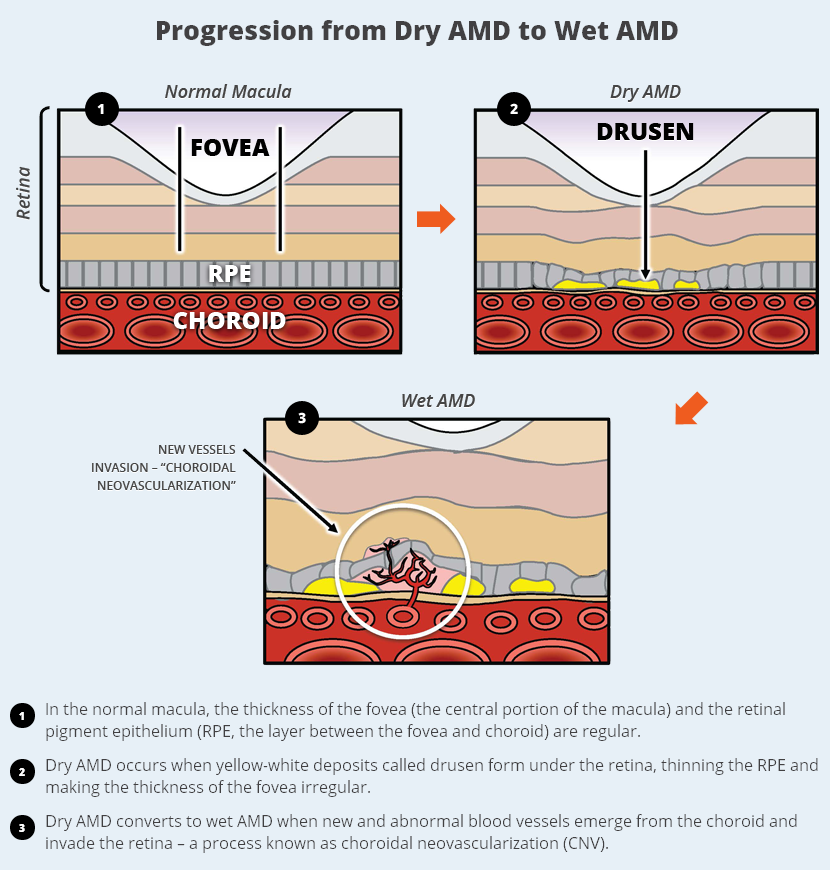
Wet AMD has no staging, it's always considered advanced. Dry AMD in some patients (even those with early stage AMD) suddenly becomes wet AMD.
Here's what Avalanche's S-1 filing says about AVA-201 (this will compete with ACT's RPE therapy using the same curative technology as AVA-101):
What is ACT's market?
We don't know yet, but Early Stage is going to be very difficult to market as there are no symptoms. Will early stage patients with no visual loss be willing to have a risky vitrectomy surgery and have hESC-derived stem cells injected into their eye? I doubt it. ACT may be able to treat some of the late-stage dry AMD market with their RPE therapy, but they are probably best suited using a yet to be developed combination therapy for this stage. Everything points to ACT primarily going after Intermediate Stage patients.
Without directly stating it, based on Avalanche's S-1 filing, with the reference to "up to 7.3 million patients at high risk of developing wet AMD," they are clearly referring to Intermediate Stage dry AMD patients.
Here are the prevalence numbers and characteristics for each stage:
Early Stage (Prevalence: [unknown] in US)
Patients have only a few small or medium-sized drusen. At this stage, vision is normal and there are no symptoms.
Intermediate Stage (Prevalence: ~7.3 million in US)*
The condition has advanced to intermediate dry AMD when the patient has many medium-sized drusen or one or more large drusen under the retina. Some of these patients notice blurring of central vision and may compensate by increasing the light when reading.
Intermediate Stage patients do not have to progress to Late Stage dry AMD to advance to Wet AMD. This assumption is a misnomer.
Late Stage (Prevalence: ~1.75 million in US; 2.95 million by 2020)*
Characterized by drusen as in the second stage, but also by a breakdown of the light-sensitive cells and supporting tissues of the macula. Patients are most likely to have blurred central vision with this stage.
*NEI statistical data, prevalence of blindness by age-group; Archives of Ophthalmology, Volume 122, April 2004
www.nei.nih.gov/eyedata/pbd_tables.asp
AMD Characteristics
www.uspharmacist.com/content/d/featured%20articles/c/27871
Prevalence of Age-Related Macular Degeneration in the US Population, JAMA Ophthalmology, Jan 2011
archopht.jamanetwork.com/article.aspx?articleid=426834
###
Interestingly, Regeneron partnered with Avalanche to develop AVA-311 to target XLRS (juvenile X-linked retinoschisis). Similar strategy to ACT targeting Stargardt's to pursue Orphan status. Regeneron purchased $10M worth of the common stock in a private placement concurrent with the IPO offering. The real goal with AVA-311 and the Regeneron deal is to develop an Ocular BioFactory platform for novel product candidates for up to 8 unnamed therapeutic targets.
AVA-322 and AVA-323, scheduled for 2015 IND, indications are "undisclosed." Any chance Schwartz and Regeneron are going after Stargardt's or MMD?

Wet AMD "Prevention" = treating Intermediate Stage dry AMD
Avalnache's Ocular BioFactory platform vs ACT's Engineered RPE ...
Ocular BioFactory Platform:
Our Ocular BioFactory platform is designed to treat the cause of ophthalmic diseases by enabling patients’ own cells to express a therapeutic protein for a sustained period of time. We use a vector derived from adeno-associated virus (AAV), which is a small, non-pathogenic virus. DNA encoding the AAV viral genes are removed and replaced with a therapeutic gene to treat a disease. The resulting vector is used to deliver and express, or transduce, the therapeutic gene to the cells of the eye to promote continuous protein production. Although AAVs are widely used for gene therapy due to their safety, stability and sustained protein expression, our Ocular BioFactory platform has distinct characteristics that provide advantages over competing gene therapy technologies using AAVs as well as other viral and non-viral vectors.
Our Ocular BioFactory platform features two key proprietary components: a novel vector screening and optimization system referred to as directed evolution, and an industrialized manufacturing process. Through directed evolution, we generate a diverse library of millions of AAV variants and subsequently screen the variants in multiple in vitro and in vivo tests to identify the optimal variant for a specific disease. Our directed evolution technology allows us to create proprietary vectors and optimize them to target cells in different layers of the retina. Each of these cell layers constitutes a potential therapeutic target for currently unmet medical needs, providing us with multiple opportunities to apply our directed evolution technology. Our industrialized manufacturing process, based on our proprietary system, is highly efficient and stable. It uses the baculovirus expression system (BVES), which is a technology for producing high levels of recombinant protein in insect-derived cells. Production yields are up to one hundred times greater than those obtained using conventional AAV production systems. Therefore, we are able to manufacture commercial grade production for large markets such as wet AMD.
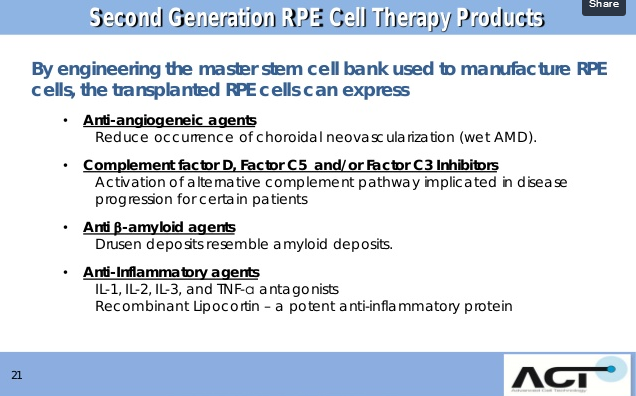
ACT's Engineered RPE Platform:
---
Given all of the above, can this explain why Schwartz is apparently the absentee representative of ACT at conferences?
Would an apparent conflict-of-interest influence and/or jeopardize the review and timely release of ACT's published results?
Here's a summary of findings ...
Avalanche's lead candidate, AVA-101, is in a Phase 2a trial targeting Wet-AMD. Results look very promising (AAVL stock has soared since IPO). Top-line results are expected mid-2015 (here's a glimpse at data presented at Euretina 2014). However, AVA-201, in pre-clinical development, is targeting Wet AMD "Prevention." And if AVA-101 is a home run, which it looks like it may be, AVA-201 could see similar success.
Per Avalanche's website:
AVA-201 is a next generation anti-VEGF gene therapy product candidate, which is being developing for the prevention of wet AMD. AVA-201 delivers the same sFLT-1 expressing gene as AVA-101 but uses next-generation AAV vector delivery method. AVA-201 is administered by an intravitreal injection directly into the vitreous, the jellylike substance inside the eye.
Viral genes are removed from AAV and replaced with specific genes encoding a therapeutic protein that specifically treats AMD. AAV can infect a variety of retinal cell types and remain stable, resulting in long-term therapeutic protein expression. In clinical studies, AAV appears to be safe and well-tolerated when injected into the retina.
We believe a novel therapy that represents a functional cure is meaningfully differentiated from standard-of-care therapies and will have access to the entire wet AMD market, not just the 40% treated with on-label Lucentis and EYLEA, but also the large pool of patients currently using Avastin off-label.
AVA-101 is designed to be administered via a single subretinal injection. The vector is placed into direct contact with retinal cells, which then produce sFLT-1. We believe that a majority of vitreoretinal surgeons are capable of performing the procedure to deliver AVA-101, which is an outpatient procedure performed under local anesthesia.
Based on data from preclinical studies, AVA-101 reaches a therapeutically beneficial level of continuous expression of sFLT-1 within six to eight weeks from administration. In animal models, AVA-101 expression has been shown to last up to 17 months, and data from other studies with AAV in the retina have shown gene expression to last more than five years. In humans, AVA-101 has been studied up to one year, and we believe it has the potential to last much longer.
AVA-101 is designed to be administered via a single subretinal injection. The vector is placed into direct contact with retinal cells, which then produce sFLT-1. We believe that a majority of vitreoretinal surgeons are capable of performing the procedure to deliver AVA-101, which is an outpatient procedure performed under local anesthesia.
Based on data from preclinical studies, AVA-101 reaches a therapeutically beneficial level of continuous expression of sFLT-1 within six to eight weeks from administration. In animal models, AVA-101 expression has been shown to last up to 17 months, and data from other studies with AAV in the retina have shown gene expression to last more than five years. In humans, AVA-101 has been studied up to one year, and we believe it has the potential to last much longer.
Wet AMD has no staging, it's always considered advanced. Dry AMD in some patients (even those with early stage AMD) suddenly becomes wet AMD.
Here's what Avalanche's S-1 filing says about AVA-201 (this will compete with ACT's RPE therapy using the same curative technology as AVA-101):
AVA-201 produces the same anti-VEGF protein as AVA-101 using a proprietary, customized delivery mechanism, or vector, that can be administered earlier in the disease progression and before the onset of wet AMD. Up to 7.3 million patients in the United States are at high risk of developing wet AMD, and we believe that the highest risk patients can be identified through a combination of clinical and genetic biomarkers. Avalanche owns exclusive rights to develop and commercialize AVA-201 worldwide.
We don't know yet, but Early Stage is going to be very difficult to market as there are no symptoms. Will early stage patients with no visual loss be willing to have a risky vitrectomy surgery and have hESC-derived stem cells injected into their eye? I doubt it. ACT may be able to treat some of the late-stage dry AMD market with their RPE therapy, but they are probably best suited using a yet to be developed combination therapy for this stage. Everything points to ACT primarily going after Intermediate Stage patients.
Without directly stating it, based on Avalanche's S-1 filing, with the reference to "up to 7.3 million patients at high risk of developing wet AMD," they are clearly referring to Intermediate Stage dry AMD patients.
Here are the prevalence numbers and characteristics for each stage:
Early Stage (Prevalence: [unknown] in US)
Patients have only a few small or medium-sized drusen. At this stage, vision is normal and there are no symptoms.
Intermediate Stage (Prevalence: ~7.3 million in US)*
The condition has advanced to intermediate dry AMD when the patient has many medium-sized drusen or one or more large drusen under the retina. Some of these patients notice blurring of central vision and may compensate by increasing the light when reading.
Intermediate Stage patients do not have to progress to Late Stage dry AMD to advance to Wet AMD. This assumption is a misnomer.
Late Stage (Prevalence: ~1.75 million in US; 2.95 million by 2020)*
Characterized by drusen as in the second stage, but also by a breakdown of the light-sensitive cells and supporting tissues of the macula. Patients are most likely to have blurred central vision with this stage.
*NEI statistical data, prevalence of blindness by age-group; Archives of Ophthalmology, Volume 122, April 2004
www.nei.nih.gov/eyedata/pbd_tables.asp
AMD Characteristics
www.uspharmacist.com/content/d/featured%20articles/c/27871
Prevalence of Age-Related Macular Degeneration in the US Population, JAMA Ophthalmology, Jan 2011
archopht.jamanetwork.com/article.aspx?articleid=426834
###
Interestingly, Regeneron partnered with Avalanche to develop AVA-311 to target XLRS (juvenile X-linked retinoschisis). Similar strategy to ACT targeting Stargardt's to pursue Orphan status. Regeneron purchased $10M worth of the common stock in a private placement concurrent with the IPO offering. The real goal with AVA-311 and the Regeneron deal is to develop an Ocular BioFactory platform for novel product candidates for up to 8 unnamed therapeutic targets.
AVA-322 and AVA-323, scheduled for 2015 IND, indications are "undisclosed." Any chance Schwartz and Regeneron are going after Stargardt's or MMD?

Wet AMD "Prevention" = treating Intermediate Stage dry AMD
Avalnache's Ocular BioFactory platform vs ACT's Engineered RPE ...
Ocular BioFactory Platform:
Our Ocular BioFactory platform is designed to treat the cause of ophthalmic diseases by enabling patients’ own cells to express a therapeutic protein for a sustained period of time. We use a vector derived from adeno-associated virus (AAV), which is a small, non-pathogenic virus. DNA encoding the AAV viral genes are removed and replaced with a therapeutic gene to treat a disease. The resulting vector is used to deliver and express, or transduce, the therapeutic gene to the cells of the eye to promote continuous protein production. Although AAVs are widely used for gene therapy due to their safety, stability and sustained protein expression, our Ocular BioFactory platform has distinct characteristics that provide advantages over competing gene therapy technologies using AAVs as well as other viral and non-viral vectors.
Our Ocular BioFactory platform features two key proprietary components: a novel vector screening and optimization system referred to as directed evolution, and an industrialized manufacturing process. Through directed evolution, we generate a diverse library of millions of AAV variants and subsequently screen the variants in multiple in vitro and in vivo tests to identify the optimal variant for a specific disease. Our directed evolution technology allows us to create proprietary vectors and optimize them to target cells in different layers of the retina. Each of these cell layers constitutes a potential therapeutic target for currently unmet medical needs, providing us with multiple opportunities to apply our directed evolution technology. Our industrialized manufacturing process, based on our proprietary system, is highly efficient and stable. It uses the baculovirus expression system (BVES), which is a technology for producing high levels of recombinant protein in insect-derived cells. Production yields are up to one hundred times greater than those obtained using conventional AAV production systems. Therefore, we are able to manufacture commercial grade production for large markets such as wet AMD.
ACT's Engineered RPE Platform:

---
Given all of the above, can this explain why Schwartz is apparently the absentee representative of ACT at conferences?
Would an apparent conflict-of-interest influence and/or jeopardize the review and timely release of ACT's published results?