|
|
Post by bernstein on Aug 5, 2016 10:54:31 GMT
IGXT (MC $42 M) 2 FDA approvals coming + 2 new NDA Filings in 4Q = 1000% Potential Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there are another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .Their unique Thin-film version of the Blockbuster Drug Cialis (Erectile Dysfunction) alone is worth $2-3 per share , NDA filing for the Drug coming before Year end . IGXT has a ridiculous Market Cap of $42 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL Intelgenx (IGXT) Market Cap: $42 Million Cash: $2.1 Million Price: $0.66 Burn-Rate: ~$300k per Quarter Shares Out : 63.6 Million Presentation July 2016 s2.q4cdn.com/790425727/files/doc_presentations/2016/INTELGENX_July-6_2016-%281%29.pdfUpcoming Milestones : Big Partnership imminent : •Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016 Product :Rizaport (Migraine) •European Mktg Approval–November 2015 •Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis •505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017Product :Indicated for Opioid Dependence •Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.IntelGenx Corp., today announced the recent initiation of a phase 1 clinical trial of montelukast, a unique drug repurposing opportunity for the treatment of degenerative diseases of the brain, such as: mild cognitive impairment and Alzheimers disease, the most prominent form of dementia. IntelGenx expects results from the phase 1 trial to be available in September 2016.  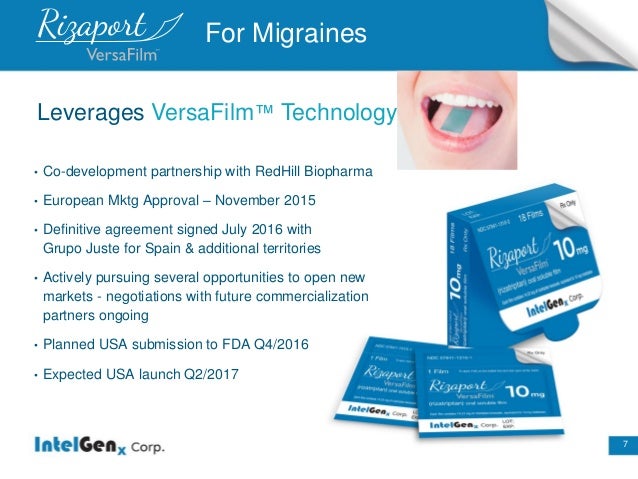  |
|
|
|
Post by bernstein on Aug 7, 2016 14:18:35 GMT
Some Infos about 2 Big Drugs of this undiscovered and brutally undervalued Goldmine ... IntelGenx Corp., today announced the recent initiation of a phase 1 clinical trial of montelukast, a unique drug repurposing opportunity for the treatment of degenerative diseases of the brain, such as: mild cognitive impairment and Alzheimers disease, the most prominent form of dementia. IntelGenx expects results from the phase 1 trial to be available in September 2016.The wonder pill that could STOP your brain ageing: Common asthma drug may be a cure for Alzheimer's, claims study..www.dailymail.co.uk/sciencetech/article-3290482/The-wonder-pill-STOP-ageing-Common-asthma-drug-cure-Alzheimer-s-claims-study.html---- Product :Tadalafil (Erectile Dysfunction) •505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017Wafer Viagra For Instant Results One of the big challenges for erectile dysfunction medicine manufacturers is to quicken the time between the application of the medicine and its response. Men want instant results when they take Viagra, Cialis or Levitra and complain that having to wait as much as thirty minutes before the active ingredient is effective can burst the bubble of spontaneity and passion which are often essential requirements of good sex. Well a solution could be at hand following research done by a Canadian research company called IntelGenx who have developed wafers [like the common breath freshness ones] that are designed to melt on your tongue which they say will get the medicine into your bloodstream around 30% faster than by using conventional tablets. IntelGenx say that because the mouth is lined with tiny blood vessels applying the wafer Viagra here can give a very fast application of the drug directly into the bloodstream. IntelGenx said that they would be conducting more clinical trials to corroborate this and they hoped to develop a whole range of fast acting wafer medications to treat not only erectile dysfunction but also pain relief, depression and hypertension. Sometimes men complain that if they have eaten a large meal it can take an even longer time for the Viagra, Cialis and Levitra to take effect. If on the other hand they made use of erectile dysfunction medication in a wafer form applied directly to the mouth and therefore bypassing their digestive system this problem would disappear. Using a wafer type of Viagra would also mean that you wouldn't have to go and get a glass of water in the middle of a passionate encounter in order to swallow your pill. Wafer Viagra would be a much more discreet application of impotence medication and Ukmedix News will be looking out for updates on the IntelGenx clinical testing done on this new idea |
|
|
|
Post by JHam on Aug 8, 2016 9:13:58 GMT
Some Infos about 2 Big Drugs of this undiscovered and brutally undervalued Goldmine ... IntelGenx Corp., today announced the recent initiation of a phase 1 clinical trial of montelukast, a unique drug repurposing opportunity for the treatment of degenerative diseases of the brain, such as: mild cognitive impairment and Alzheimers disease, the most prominent form of dementia. IntelGenx expects results from the phase 1 trial to be available in September 2016.The wonder pill that could STOP your brain ageing: Common asthma drug may be a cure for Alzheimer's, claims study..www.dailymail.co.uk/sciencetech/article-3290482/The-wonder-pill-STOP-ageing-Common-asthma-drug-cure-Alzheimer-s-claims-study.html---- Product :Tadalafil (Erectile Dysfunction) •505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017Wafer Viagra For Instant Results One of the big challenges for erectile dysfunction medicine manufacturers is to quicken the time between the application of the medicine and its response. Men want instant results when they take Viagra, Cialis or Levitra and complain that having to wait as much as thirty minutes before the active ingredient is effective can burst the bubble of spontaneity and passion which are often essential requirements of good sex. Well a solution could be at hand following research done by a Canadian research company called IntelGenx who have developed wafers [like the common breath freshness ones] that are designed to melt on your tongue which they say will get the medicine into your bloodstream around 30% faster than by using conventional tablets. IntelGenx say that because the mouth is lined with tiny blood vessels applying the wafer Viagra here can give a very fast application of the drug directly into the bloodstream. IntelGenx said that they would be conducting more clinical trials to corroborate this and they hoped to develop a whole range of fast acting wafer medications to treat not only erectile dysfunction but also pain relief, depression and hypertension. Sometimes men complain that if they have eaten a large meal it can take an even longer time for the Viagra, Cialis and Levitra to take effect. If on the other hand they made use of erectile dysfunction medication in a wafer form applied directly to the mouth and therefore bypassing their digestive system this problem would disappear. Using a wafer type of Viagra would also mean that you wouldn't have to go and get a glass of water in the middle of a passionate encounter in order to swallow your pill. Wafer Viagra would be a much more discreet application of impotence medication and Ukmedix News will be looking out for updates on the IntelGenx clinical testing done on this new idea Thanks, I'll take a closer look. The think that jumps right out at me is the $2.1M in cash. I know they have a very low burn rate, but that's still not much. I also see that the FDA rejected their first attempt to get Versafilm approved in 2014. Has that been resolved or is that what they are expecting now? Just asking things that jumped out at me after a very quick glance. Thanks. |
|
|
|
Post by bernstein on Aug 8, 2016 11:28:16 GMT
Hi Here is News that comes Friday after hour , they have now enough cash to drive the company into profitability which could happen next year . IntelGenx Announces Sale of Forfivo XL(R) Royalty for $6 Million....August 05, 2016 www.intelgenx.com/investors/press-releases/press-release-details/2016/IntelGenx-Announces-Sale-of-Forfivo-XLR-Royalty-for-6-Million/default.aspxThe Drug that was rejected by FDA back in 2014 is the Drug Rizaport below the issues are resolved and Resubmission is expected next Quarter . Product :Rizaport (Migraine) •European Mktg Approval–November 2015 •Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017ir.redhillbio.com/releasedetail.cfm?releaseid=823064FDA's letter accepted the bioequivalence study and safety information submitted and requires no additional clinical studies; IntelGenx and RedHill plan to address remaining issues, primarily related to third party manufacturing, packaging and labeling, within weeks based on available data |
|
|
|
Post by bernstein on Aug 9, 2016 11:23:27 GMT
|
|
|
|
Post by bernstein on Aug 12, 2016 13:21:10 GMT
The Company has now Cash untill mid 2018 excluding Upfront Payments from new Partnerships and Product Revenues . Partnerdeal news and Phase 1 Results of their Alzheimer Drug coming within 6 Weeks . Market Cap still way below $50 Million which is still a big Joke . from yesterdays 2Q Results www.intelgenx.com/investors/press-releases/press-release-details/2016/IntelGenx-Reports-Second-Quarter-2016-Financial-Results-and-Appointment-of-New-Director/default.aspxContinued late-stage discussions with global pharmaceutical companies for multiple products with the potential goal of concluding a definitive agreement to be finalized in the third quarter of 2016Initiated a phase 1 study of Montelukast for the treatment of degenerative diseases of the brain, such as: mild cognitive impairment and Alzheimers disease, the most prominent form of dementia. IntelGenx expects results from the phase 1 trial to be available in September 2016. |
|
|
|
Post by bernstein on Aug 16, 2016 13:06:46 GMT
A lot of new Partnerdeals on the way which means Upfront Payments to the Company . Market Cap of $43 Million is still laughable for this Goldmine .GL Transcript Q2 released last Thursday s2.q4cdn.com/790425727/files/doc_financials/2016/final-081116-intelgenx-q2-2016-financial-results-47950179-v.3.pdfWith respect to other BD activities, we're clearly focusing right now in the business development area on bringing in new deals. For example at the recent BIO Conference, we met with 50 different pharmaceutical companies to discuss partnering opportunities, and we're currently in discussions with eight companies that are interested in RIZAPORT licenses, 17 companies that are interested in our Tadalafil product, three companies are interested in our schizophrenia drug, and five companies have expressed interest in a deal on Forfivo outside of the US. Above and beyond that, there are ongoing late-stage discussions with a global pharmaceutical company for multiple products with the potential goal of finalizing a definitive agreement in the third quarter of 2016. |
|
|
|
Post by bernstein on Aug 18, 2016 14:45:21 GMT
|
|
|
|
Post by bernstein on Aug 19, 2016 18:50:08 GMT
|
|
|
|
Post by bernstein on Aug 22, 2016 13:08:24 GMT
GREAT NEWS OUT .. and stock is stiill terrible underpriced at a market cap of laughable $52 Million .GL www.intelgenx.com/investors/press-releases/press-release-details/2016/IntelGenx-Announces-Successful-Clinical-Study-for-Montelukast-for-the-Treatment-of-Degenerative-Diseases-of-the-Brain/default.aspxIntelGenx Announces Successful Clinical Study for Montelukast for the Treatment of Degenerative Diseases of the Brain August 22, 2016 "We are very pleased by the results of the study which confirmed that our Montelukast VersaFilm™ significantly improves the bioavailability of the drug when given as an oral film product. The study demonstrates that the VersaFilm™ technology is an effective delivery system for CNS applications," said Dr. Horst G. Zerbe, President and CEO of IntelGenx. "Montelukast is an important drug repurposing opportunity that the company has in its product pipeline which could potentially become a multi-billion-dollar product in the large and underserved market for degenerative diseases of the brain." |
|
|
|
Post by bernstein on Aug 24, 2016 16:44:21 GMT
|
|
|
|
Post by CM kipper007 on Aug 25, 2016 17:30:24 GMT
Have you heard anything about them wanting to change exchange?
|
|
|
|
Post by bernstein on Aug 26, 2016 13:34:42 GMT
They work on it but at this time they doesnt meet the requirement for Uplisting to Nasdaq . New Presenation out shows that there is already strong interest in Montelukast after positive results (page 13) .We have a MEGA Pipeline ,a lot of Cash for 2 Years and many Drugs close to NDA Submissions all this for a brutal terrible low valuation of $50 Million .IGXT is definitely a MUST HAVE at these levels and patience will payoff big time here thats for sure . s2.q4cdn.com/790425727/files/doc_presentations/2016/IGXT-Aug-22-Investor-Presentation.pdf |
|
|
|
Post by bernstein on Aug 29, 2016 14:38:11 GMT
|
|
|
|
Post by bernstein on Aug 30, 2016 15:04:02 GMT
|
|
|
|
Post by bernstein on Aug 31, 2016 14:15:02 GMT
Looking great again could hit $1 today then real run will begin ... Market cap of $58 Million is still terrible undervalued its a big joke .
|
|
|
|
Post by CM kipper007 on Aug 31, 2016 17:13:40 GMT
Congrats Bernstein!
|
|
|
|
Post by actcrazy on Aug 31, 2016 21:09:25 GMT
Thanks for the dd Bernstein. I bought a good chunk for fun at seventy cents a few days back and ...so far ...much happy. Massive volume today, near the end of session shorts worked hard at bringing it down. Good luck to all here.
|
|
|
|
Post by bernstein on Sept 1, 2016 17:57:49 GMT
Thanks Guys .. The heavy MM manipulation today created another great buying opportunity . Partner deal news which is expected within this month will push the stock much much higher .
|
|
|
|
Post by CM kipper007 on Sept 7, 2016 16:45:27 GMT
sports.yahoo.com/news/intelgenx-present-18th-annual-rodman-135252093.htmlSAINT-LAURENT, CANADA--(Marketwired - Sep 7, 2016) - IntelGenx Corp., (TSX VENTURE:IGX) (IGXT), today announced that Dr. Horst Zerbe, President and Chief Executive Officer, will present at two upcoming conferences. Also present and available for questions will be Andre Godin, Executive Vice-President and Chief Financial Officer. Rodman & Renshaw 18th Annual Global Investment Conference Date: Monday, September 12 Location: Lotte New York Palace Hotel, New York City Presentation Time: 10:50 AM Eastern Time 11th Annual Singular Research Conference "Best of the Uncovereds" Date: Thursday, September 22 Location: Luxe Sunset Boulevard Hotel, Los Angeles About IntelGenx: IntelGenx is a leading oral drug delivery company focused on the development and manufacturing of innovative pharmaceutical oral films based on its proprietary VersaFilm™ technology platform. Established in 2003, the Montreal-based company is listed on the TSX-V and OTC-QX. IntelGenx highly skilled team provides comprehensive pharmaceuticals services to pharmaceutical partners, including R&D, analytical method development, clinical monitoring, IP and regulatory services. IntelGenx state-of-the art manufacturing facility, established for the VersaFilm™ technology platform, supports lab-scale to pilot and commercial-scale production, offering full service capabilities to our clients. More information is available about the company at: www.intelgenx.com. Forward Looking Statements: This document may contain forward-looking information about IntelGenx' operating results and business prospects that involve substantial risks and uncertainties. Statements that are not purely historical are forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. These statements include, but are not limited to, statements about IntelGenx' plans, objectives, expectations, strategies, intentions or other characterizations of future events or circumstances and are generally identified by the words "may," "expects," "anticipates," "intends," "plans," "believes," "seeks," "estimates," "could," "would," and similar expressions. All forward looking statements are expressly qualified in their entirety by this cautionary statement. Because these forward-looking statements are subject to a number of risks and uncertainties, IntelGenx' actual results could differ materially from those expressed or implied by these forward looking statements. Factors that could cause or contribute to such differences include, but are not limited to, those discussed under the heading "Risk Factors" in IntelGenx' annual report on Form 10-K, filed with the United States Securities and Exchange Commission and available at www.sec.gov, and also filed with Canadian securities regulatory authorities and www.sedar.com. IntelGenx assumes no obligation to update any such forward-looking statements. Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange), nor the OTCQX accepts responsibility for the adequacy or accuracy of this release. Source: IntelGenx Technologies Corp.
|
|










