|
|
Post by bernstein on Sept 20, 2017 15:35:53 GMT
MDNA.TO (MC C$36 M) (Cash C$20 M) Hot Cancer Play with important Results in Q1 that could lead to FDA approval =1500-2000% UPSIDE Potential ! Brutally cheap and completely unknown canadian biotech stock with minimum 10 bagger potential . Positive Phase 2 results in recurrent glioblastoma which expected in Q1 2018 could lead to accelerated approval which would be a HUGE MAJOR milestones for this undiscovered low float stock . This company has brutally low valuation of only $36 million and most of that in cash $20 million which is enough untill Q1 2019 . This stock at $1.50 is a lifetime opportunity and we could see $15-20 easily on Positive 2 results and FDA approval .GL Medicenna Therapeutics (MDNA.TO) Market Cap C$36 Million Cash : C$ 20 Million << enough untill Q1 2019 Price : $1.50 Shares Out: 24.3 Million ( 15.5 Million shares held by Insider alone ) Medicenna Therapeutics (TSXV: MDNA) CEO: Our Drug Has Potential for Phase 2 Approval smallcappower.com/videos/companies-to-watch/medicenna-therapeutics-tsxv-mdna/New Presentation s21.q4cdn.com/710416940/files/doc_presentations/2017/Medicenna-Corporate-Presentation-Q3-2017.pdfFact Sheet s21.q4cdn.com/710416940/files/doc_downloads/fact_sheet/Medicenna-FS-Q1-2017-v2.pdfInsider Ownership : Aries Biologics, Inc. 5 500 000 Rosemina Merchant 5 050 000 Fahar Merchant 5 050 000 Elizabeth Williams 5 300 Chandrakant J. Panchal, PhD 1 000 Trevor P. Wong-Chor 714 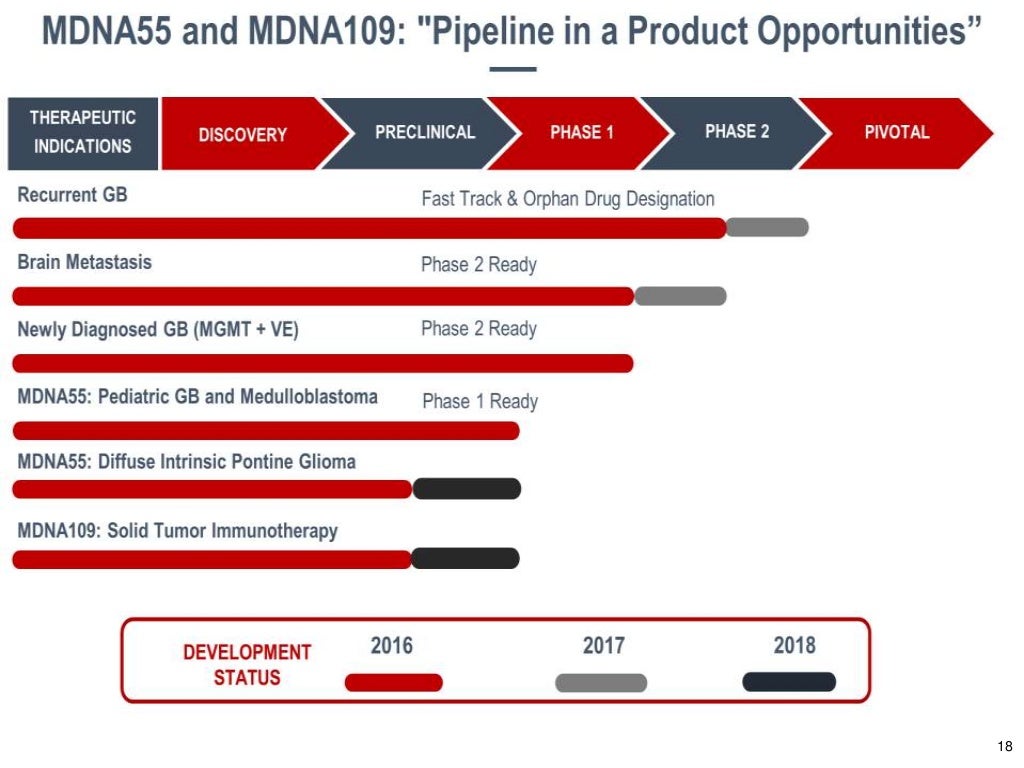 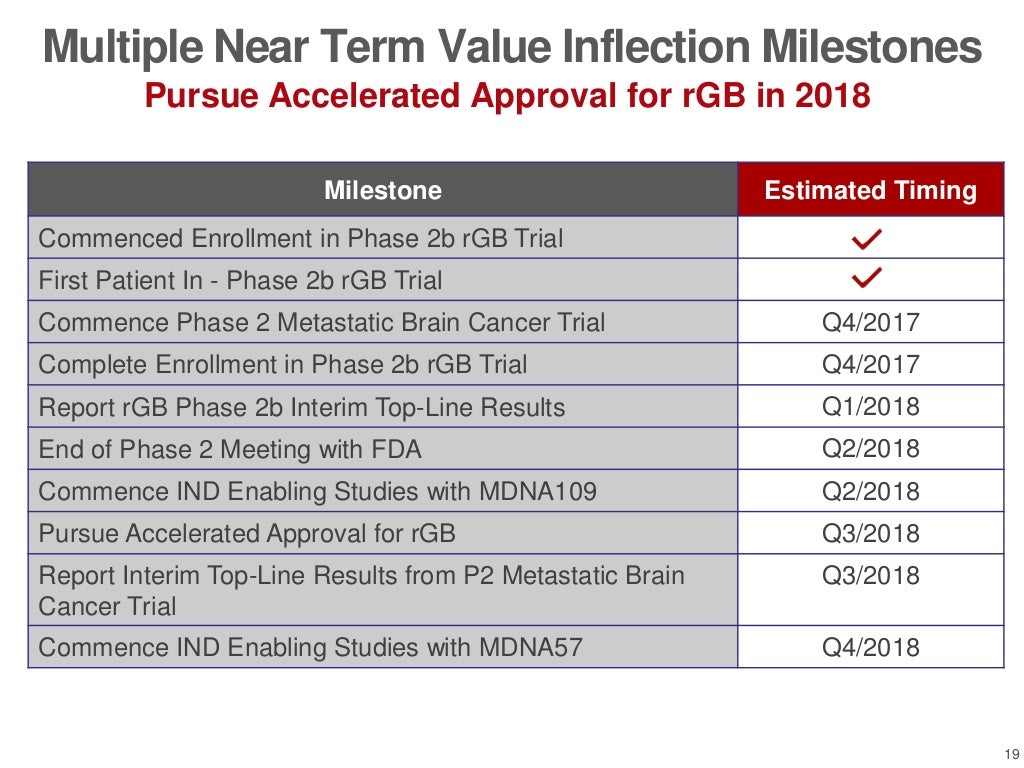  |
|
|
|
Post by bernstein on Sept 21, 2017 14:24:05 GMT
Small Phase 2b trial has huge chance to be successful because it was already successfull in prior studies so this stock like to be above $10 after phase 2 results in early Q1 . And that Management holding 15+ million shares of the 24.3 million shares outstanding makes me also optimistic .GL MDNA55 is enrolling patients in a Phase 2b clinical trial for rGB at leading brain cancer centres in the US. MDNA55 has completed 3 clinical trials in 72 patients, including 66 adults with rGB, demonstrated compelling efficacy and obtained Fast-Track and Orphan Drug status from USFDA. Unlike most other cancer therapies, Medicenna's IL4-ECs have the potential to purge both the tumor and the immunosuppressive tumor microenvironment, Medicenna Phase 2b trial using Brainlab technology www.biotuesdays.com/briefs/2017/6/8/medicenna-phase-2b-trial-using-brainlab-technologyThe current Phase 2b clinical trial plans to enroll 43 adult patients with rGB at leading brain cancer centers in the U.S. |
|